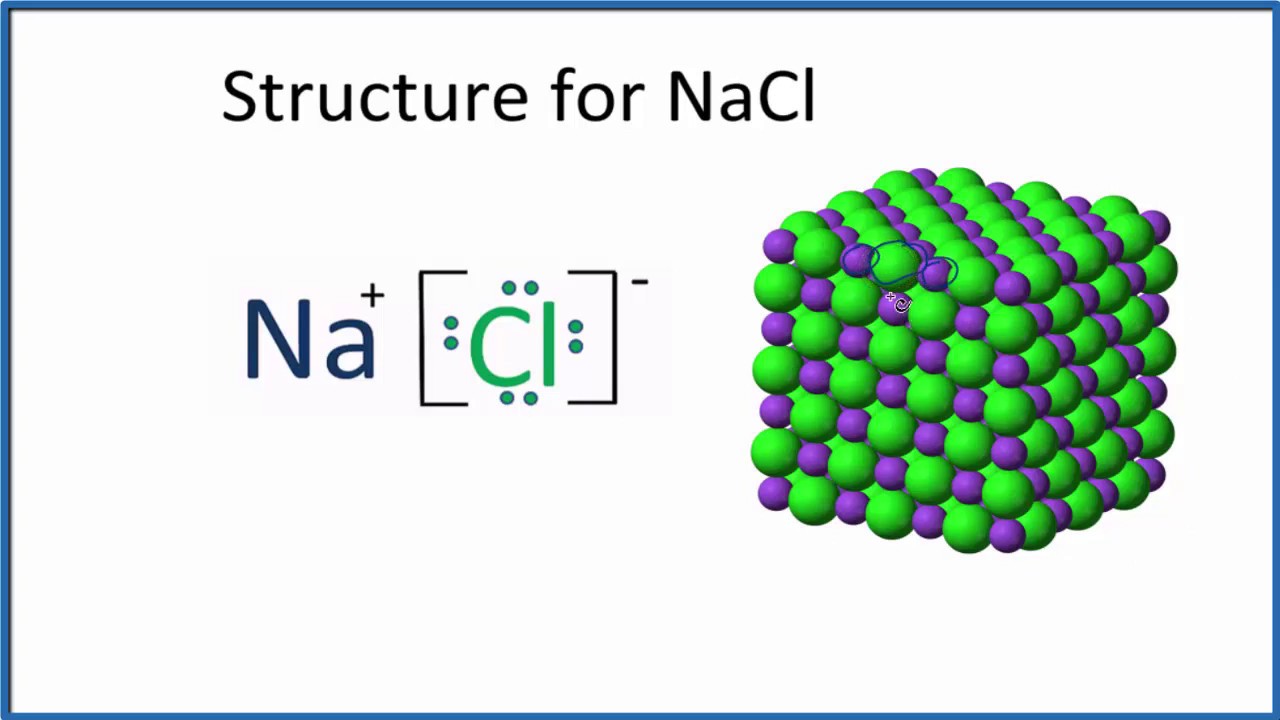 
The others are progressively more insoluble in water (K sp is 10 -10, 10 -13, and 10 -16 for AgCl, AgBr, and AgI), reflecting increasing covalency as Δχ decreases. Of these compounds, only AgF is soluble in water and should be thought of as an ionic compound. Science Chemistry 8.83 Construct a Born-Haber cycle for the formation of the hy- pothetical compound NaCl2, where the sodium ion has a 2+ charge (the second ionization energy for sodium is given in Table 7.2). Our lattice energy calculation overestimates the ionic contribution in the case of the heavier silver halides, but underestimates the covalent contribution. For example, the lattice energy of sodium chloride crystal is the energy change of reaction when Na and Cl ions react to produce a NaCl crystal, which is equal to -786 kJ/mol. The covalent bonding contribution to the lattice energies of AgCl, AgBr, and AgI makes these salts sparingly soluble in water.Īgain, we can interpret the fortuitous agreement between the calculated and experimentally obtained energies in terms of compensating errors. Lattice energy is the heat of formation when one mole of crystalline ionic solid is generated from its constituent ions in their gaseous state. This reaction is used as a qualitative test for the presence of halide ions in solutions. Should we interpret the good agreement with values calculated from the ionic model to mean that these compounds are ionic? Clearly, this description is inappropriate for AgI, where the electronegativity difference Δχ is only 0.6 (compare this value to 0.4 for a C-H bond, which we typically view as non-polar).Ī drop of siver nitrate solution, when added to a dilute hydrochloric acid solution, results in the immediate formation of a white silver chloride precipitate. However we are still obtaining answers within about 12% error even for AgI. Looking at the table, we see that the error is small for AgF and becomes progressively larger for the heavier silver halides. It is interesting to repeat this exercise for the silver halides, which have either the NaCl structure (AgF, AgCl, AgBr) or zincblende structure (AgI). 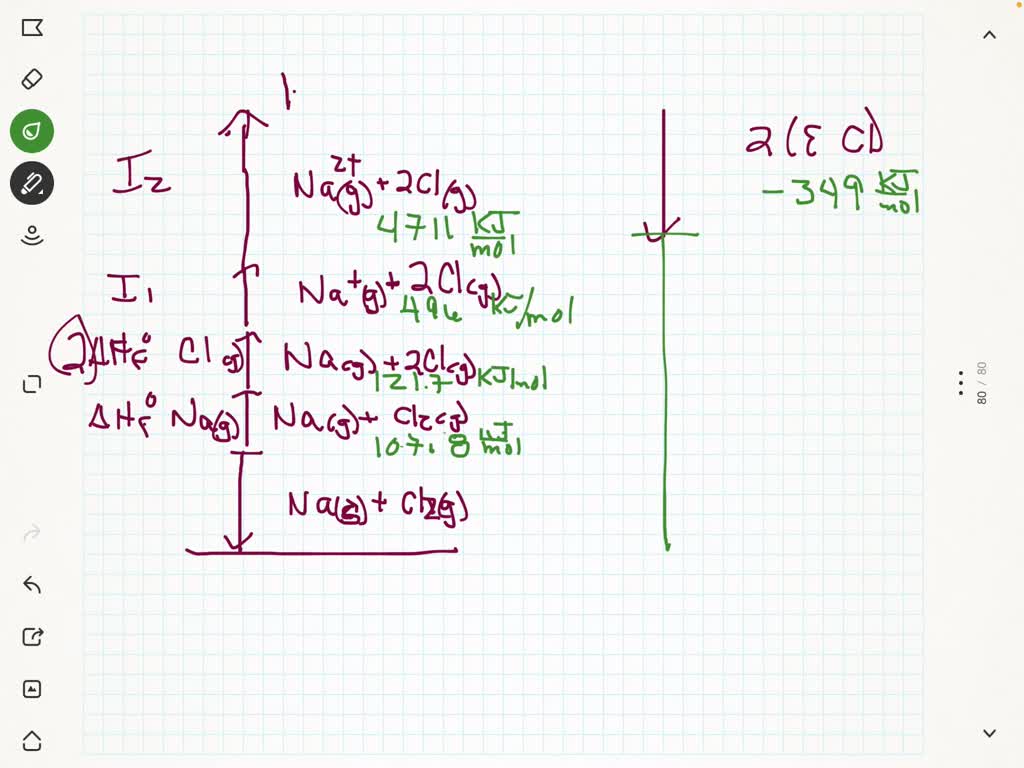
The errors in this case are only about 1% of E L. The table below shows results of more detailed lattice energy calculations for ionic fluorides in which the van der Waals term is explicitly included. We can do better by explicitly including the short-range van der Waals attractive energy between ions. The two errors partially compensate, so the overall error in the calculation is small. The strong electrostatic attraction between Na + and Cl ions holds them tightly together in solid NaCl. This expression takes into account both the Born interactions as well as the Coulomb attractions. If we underestimate the attractive energy of the crystal lattice, the energy minimization criterion ensures that the repulsion energy is underestimated as well. The Born-Landé equation is a concept originally formulated in 1918 by the scientists Born and Landé and is used to calculate the lattice energy (measure of the strength of bonds) of a compound. This is because we used energy minimization to obtain the repulsion energy in the Born-Mayer equation. The result is promising because we neglected the van der Waals term.īut.how did we get away with neglecting the van der Waals term? Here we have to subtract 2RT to convert our cycle of energies to a cycle of enthalpies, because we are compressing two moles of gas in making NaCl(s) and PΔV = ΔnRT, where Δn = -2.Įxperimentally ΔH f for NaCl is -411 kJ/molīecause all the other numbers in the cycle are known accurately, the error in our calculation is only about 15 kJ (about 2% of E L). 
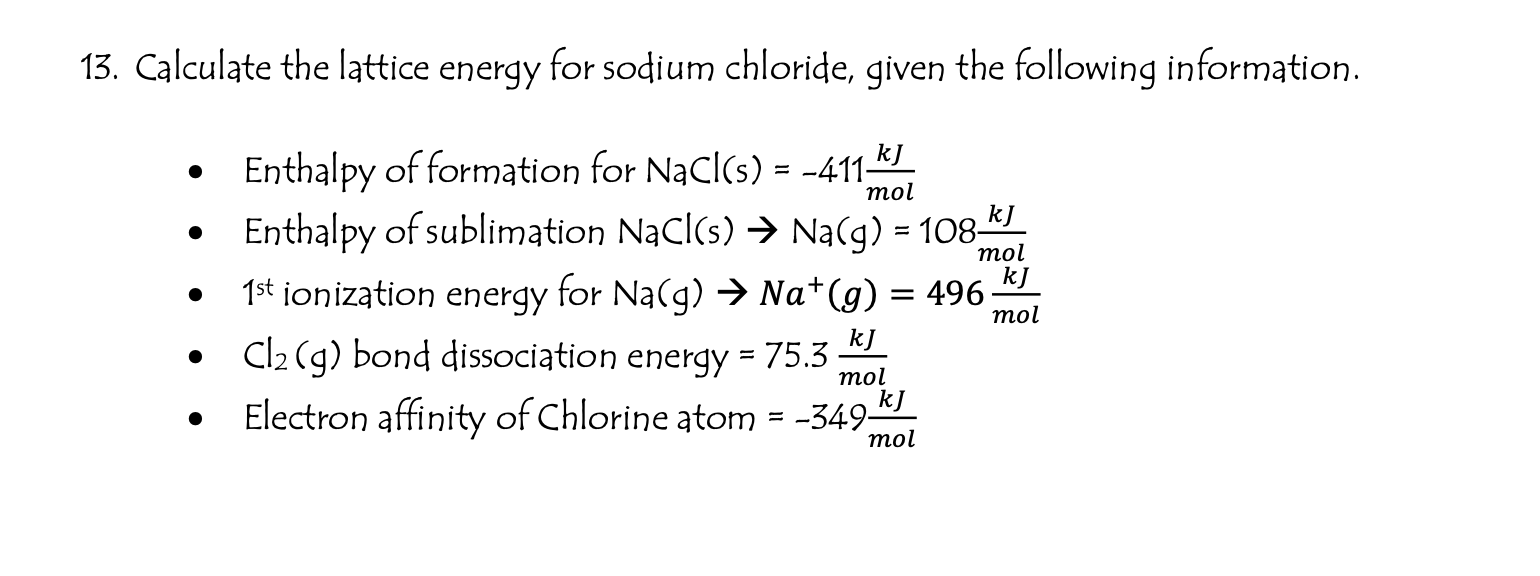
Then, the elements are converted into gaseous ions, and lastly, they are converted into a solid lattice.įigure 4 shows the basic diagram of the Born-Haber cycle for the reaction of a metal (X) with a diatomic halogen (X 2).\) So, the cycle always starts with the elements in their standard state. This calculator should be used in conjunction with the notes on Understanding Crystal Structures. The Born-Haber cycle assumes that the final product is the solid ionic compound. The Born-Haber cycle is a method used to calculate the lattice energy or Enthalpy of Formation of ionic compounds formed from gaseous ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
